Computational Thermodynamics
← Back to ResearchPhase diagrams, or equilibrium diagrams, depict the stability regions of phases in material systems under equilibrium conditions based on thermodynamic variables like composition, temperature, and pressure. They are critical in developing and manufacturing new alloys, helping to design heat treatment processes to achieve desired properties. Additionally, phase diagrams are essential for identifying and resolving performance issues in commercial alloys.
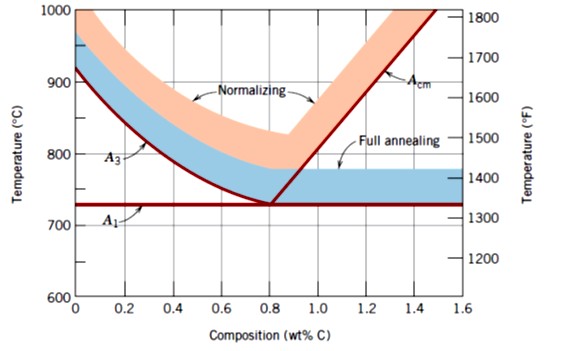
Figure 1: Austenitizing temperature for full annealing and normalizing.
Historically, phase diagrams have been determined through experimentation, which can be labor-intensive, costly, and impractical, especially for multicomponent systems with broad compositional and temperature ranges. To complement this traditional approach, theoretical calculations offer a more efficient alternative. The main advantage of this method is that by applying appropriate thermodynamic descriptions of the phases in an alloy system, information from simpler, lower-order systems can be used to predict the phase diagram of the more complex multicomponent system.
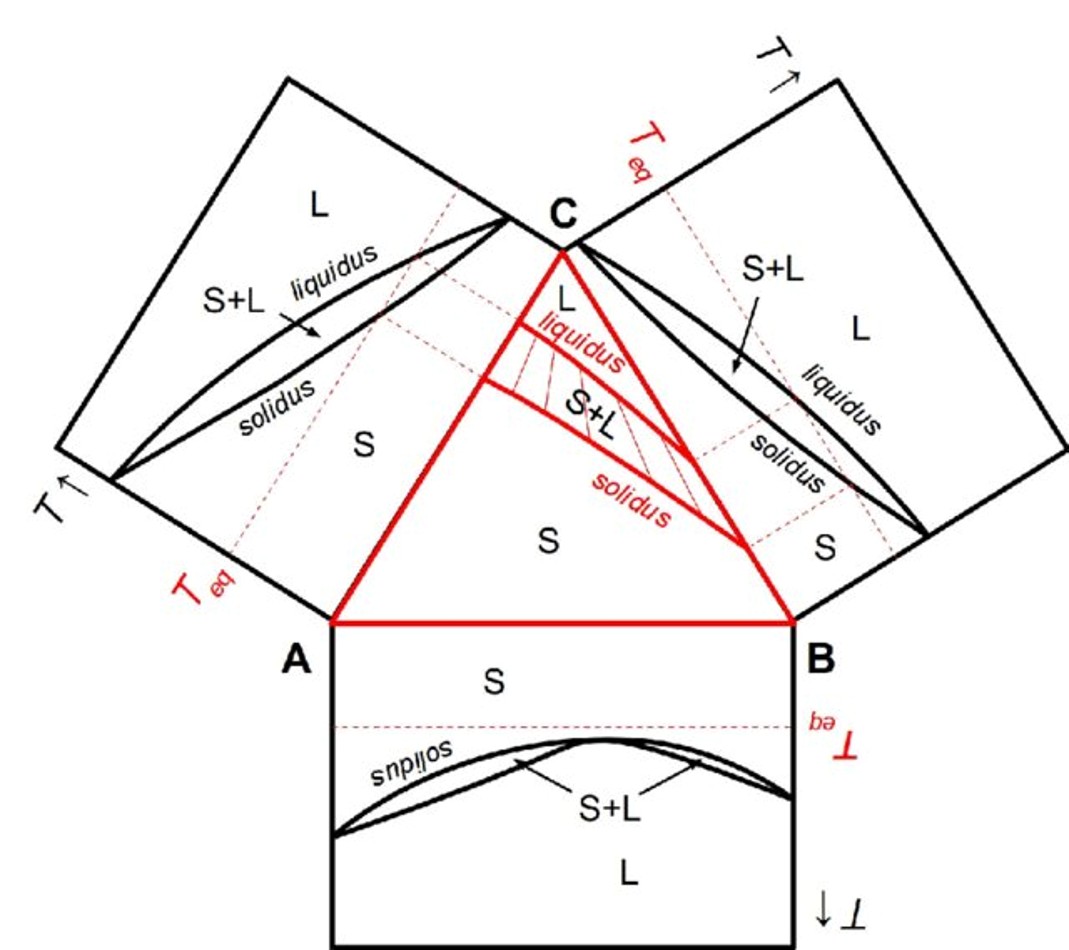
Figure 2: Computational Thermodynamics Approach
This predicted diagram can help identify specific regions of composition and temperature, where critical information can be gathered with minimal experimental effort. Once initial predictions are made, experimental data can be used to refine the interaction parameters of the multicomponent system, optimizing the data set and improving the accuracy of the phase diagram. This approach, known as Computational Thermodynamics (CT) (Kattner, 1997), enables the construction of a complete thermodynamic description of complex systems with reduced experimental work.
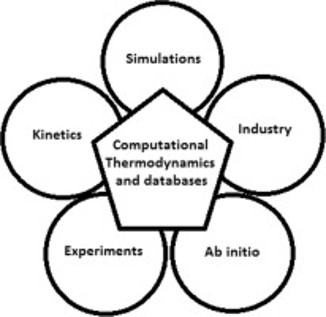
Figure 3: Applications of Computational Thermodynamics
CT Activities:
- Data: Determination of phase diagrams and thermochemical data.
- Modeling: Developing Gibbs energy models for individual phases.
- Optimization: Coupling phase equilibria and thermochemistry to create consistent thermodynamic databases for multi-phase, multicomponent systems.
- Application: Predicting thermodynamic properties and phase equilibria of multicomponent systems.